5 Energy Pyramid Examples Explained
Energy pyramid examples include; soil microbial, photosymbiotic, forest, marine, and desert energy pyramids.
This article discusses energy pyramid examples, as follows;
1). Forest Energy Pyramid (as one of the Energy Pyramid Examples)
The energy pyramid of a forest ecosystem is a pictorial or explanatory representation of bioenergy transfers in the form of feeding relationships between forest organisms of various species, and in different trophic levels of the system.
Typically, the forest energy pyramid begins from plants, which are also the primary producers or energy suppliers [5].
In forests, the role of plants is a significant one, as they make up a great portion of the total species and biomass. Plants supply energy to the forest ecosystem through photosynthesis, whereby they capture and convert solar energy to chemical bioenergy that can be easily assimilated by living organisms.
When plants grow (using the energy/food produced by photosynthesis), they become available to the primary consumers, which are unable to produce their own food, and directly dependent on plants for survival.
In a forest ecosystem, primary consumers are herbivorous animals like hares, locusts and deer. These organisms are able to receive approximately 10% of the total energy produced by plants, while the remaining 90% is waste energy that is lost as heat or in other forms.
Secondary consumers occupy the trophic level directly above primary consumers. In a forest, tertiary consumers include smaller carnivores like coyotes, some birds of prey, carnivorous plants, and even insects.
These organisms feed mainly on primary consumers, especially the smaller herbivores like hares and locusts. In many cases, the primary consumers are the only source of food for secondary consumers.
Apex predators occupy the highest trophic level in the hierarchical structure of forest energy pyramids. The apex predators in a forest ecosystem are mainly large carnivores like bears, leopards, crocodiles, and golden eagles.
It is important to note that there is little disparity between tertiary consumers and apex predators in any natural ecosystem. The two terms can be used interchangeably, although ‘tertiary consumers’ is more often used when referring to the ability of these organisms to feed on both primary and secondary consumers.
Decomposers also play an important role in forests, as these microbes (and some macro-organisms like earthworms) facilitate biodegradation of plant and animal biomass, whereby nutrients are released into the soil for plant growth.
Some organisms can function effectively at multiple trophic levels in forests. These include bears, which are omnivorous and can feed on plants [3].
Also, the trophic level of any given organism in a forest or any ecosystem depends on the size of the ecosystem, and the species that are present. For example; a coyote will occupy the apex predator level in the absence of bears.
The conditions and feeding relationships in forests are generally very similar to those that occur in other plant-dominant ecosystems, like tundras and grasslands; where vegetation serves as a major food source and shelter.

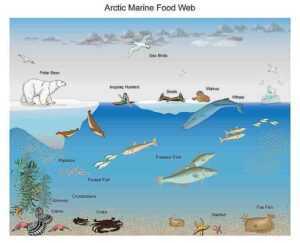
2). Marine Energy Pyramid
The marine energy pyramid represents feeding relationships and energy transfers among marine organisms of all families and species, from the level of producers to that of tertiary consumers.
Producers in a marine ecosystem include all organisms that possess chlorophyll for photosynthesis, such as seaweeds and phytoplankton.
The photosynthetic activities of primary producers in the ocean, help it to perform its function as a natural carbon sink, by capturing carbon dioxide and using it as an ingredient for the production of energy and biomass [7].
Phytoplankton are also used in ocean fertilization, which is one of the carbon removal technologies used for environmental decarbonization [4].
Primary consumers in a marine ecosystem include all organisms that rely mainly on marine plants and phytoplankton for feeding and survival; such as small fish, some crustaceans, zooplankton, sea turtles and urchins.
These organisms are prey to the marine secondary consumers, which include corals, sardines, small shark, shrimps and herring.
Tertiary consumers or apex predators in a marine ecosystem include large sharks and whales.
Feeding relationships in marine ecosystems are often complex, so that dense food webs can be traced in the ocean. These relationships are what help to establish sustainability of energy flow and nutrient circulation.
Microorganisms that decompose biomass are also present in the ocean, although the rate and pattern of biodegradation is not always identical to those of terrestrial ecosystems.
Compared to terrestrial ecosystems, the ocean is also effective in terms of energy conservation. Some nutrients like calcite in coral tests, are not always rapidly recycled, but may accumulate to form coral reefs and other geologic formations that have ecologic importance [6].
Humans may interrupt the natural flow in marine ecosystems, through fishing and exploration activities. In some cases like oil spill hazards, these interruptions may have significant biological and environmental impacts.
3). Soil Microbial Energy Pyramid (as one of the Energy Pyramid Examples)
The soil can be viewed as a robust and fully-functional ecosystem; because it serves as a habitat to a broad variety of organic species, abiotic components, and biochemical processes.
In larger ecosystems, the soil is mostly associated with the lowest trophic level, due to its role in supporting primary producers (plants) in their growth and photosynthesis.
When functioning as an ecosystem of its own, the soil also begins its energy recycling process from plants. Solar energy serves as the primary energy source which plants use as a driving-force for photosynthesis.
Not all organisms in a soil ecosystem are microbes. However, it is called ‘soil microbial energy pyramid’ due to the prominent role played by microorganisms in soil energy cycling.
Primary consumers in soil include nematodes, fungi, bacteria, and some insects. These directly depend on plants for their nutrition.
Secondary consumers in the soil ecosystem are mainly decomposers, and other organisms that feed on primary consumers; like beetles, worms, protozoans, and mites.
There is little disparity between secondary and tertiary consumers in soil. Termites are an example of soil tertiary consumers, which may feed on both primary and secondary consumers.
A healthy soil ecosystem with abundant biodiversity is essential for environmental sustainability. Optimizing soil biodiversity and health is the focus of some sustainable agricultural practices like cover cropping, mulching, composting and conservation tillage [1].
4). Desert Energy Pyramid
The desert energy pyramid receives energy from primary producers, which include grasses, cacti and other shrub species.
Primary consumers in a desert include; gazelles, camels; kangaroo rats, rabbits and butterflies; which depend directly on desert plants for food.
Desert secondary consumers include all small carnivores that are adapted to arid conditions, such as tarantulas, lizards, and snakes.
Some tertiary consumers in the desert are; sand cat, hawk, striped hyena, vulture and desert fox. These may feed on organisms from multiple lower trophic levels of the ecosystem.
Decomposers and microbes in general are also present in deserts, although their role is less prominent than in other terrestrial ecosystems.
Aside fully-developed deserts, other arid terrains and those susceptible to desertification, may exhibit similar ecologic attributes to deserts.
5). Photosymbiotic Energy Pyramid (as one of the Energy Pyramid Examples)
Photosymbiotic energy pyramids represent the sustainable energy transfer in symbiotic (mutually beneficial) feeding relationships that involve autotrophic or photosynthetic organisms.
A classic example of photosymbiosis is the feeding relationship between corals and symbiodium; a photosynthetic microalgae [2].
In photosymbiotic energy pyramids, the autotroph is the primary source of energy, which is supplied to other organism(s) in the feeding micro-ecosystem that is established.
Both macro and microorganisms can coexist and occupy various trophic levels in a photosymbitoic energy pyramid.
While algae is often a permanent component, other organisms which may occur in photosymbiotic energy pyramids are; molluscs, sponges, ascidians and cnidarians.
Conclusion
Energy pyramid examples are;
1. Forest Energy Pyramid
2. Marine Energy Pyramid
3. Soil Microbial Energy Pyramid
4. Desert Energy Pyramid
5. Photosymbiotic Energy Pyramid
References
1). Carrera, L. M.; Buyer, J.; Vinyard, B. T.; Abdul-Baki, A. A.; Sikora, L. J.; Teasdale, J. R. (2007). “Effects of cover crops, compost, and manure amendments on soil microbial community structure in tomato production systems.” Applied Soil Ecology 37(3):247-255. Available at: https://doi.org/10.1016/j.apsoil.2007.08.003. (Accessed 8 November 2022).
2). Clavijo, J. M.; Donath, A.; Serôdio, J.; Christa, G. (2017). “Polymorphic adaptations in metazoans to establish and maintain photosymbioses.” Biological reviews of the Cambridge Philosophical Society 93(4). Available at: https://doi.org/10.1111/brv.12430. (Accessed 7 November 2022).
3). Duffy, J. E.; Cardinale, B. J.; France, K.; McIntyre, P. B.; Thébault, E.; Loreau, M. (2007). “The functional role of biodiversity in ecosystems: Incorporating trophic complexity.” Ecology Letters 10(6):522-38. Available at: https://doi.org/10.1111/j.1461-0248.2007.01037.x. (Accessed 8 November 2022).
4). Harrison, D. P. (2017). “Global negative emissions capacity of ocean macronutrient fertilization.” Environmental Research Letters 12(3). Available at: https://doi.org/10.1088/1748-9326/aa5ef5. (Accessed 8 November 2022).
5). Hui, D. (2012) “Food Web: Concept and Applications.” Nature Education Knowledge 3(12):6. Available at: https://www.nature.com/scitable/knowledge/library/food-web-concept-and-applications-84077181/. (Accessed 8 November 2022).
6). Jones, N. S.; Ridgwell, A.; Hendy, E. J. (2015). “Evaluation of coral reef carbonate production models at a global scale.” Biogeosciences 12(5). Available at: https://doi.org/10.5194/bg-12-1339-2015. (Accessed 8 November 2022).
7). Lai, Q.; Ma, J.; He, F.; Zhang, A.; Pei, D.; Yu, M. (2022). “Current and Future Potential of Shellfish and Algae Mariculture Carbon Sinks in China.” International Journal of Environmental Research and Public Health 19(14):8873. Available at: https://doi.org/10.3390/ijerph19148873. (Accessed 8 November 2022).




